- Business Process Management (BPM)Document Management System (DMS)Electronic Quality Management System (QMS)Risk, Governance & Compliance (GRC)Low Code Rapid Application Development (LC)Business Continuity Management (BCM)Enterprise Architecture (EA)Business Process Management (BPM)Document Management System (DMS)
- Document Control Overview
- AI Content Creation & Improvement
- Policy & Procedure Management (SOP)
- AI Content Mining Parser
- Collaboration & Governance
- Data Migration & Integration
- Interfacing Offline App
 Electronic Quality Management System (QMS)
Electronic Quality Management System (QMS)- Quality Management System Overview
- Document Control & Records Management
- Audit & Accreditation Management
- Corrective & Preventative Action
- Quality Event (Non-conformity / Complaint/ Compliance)
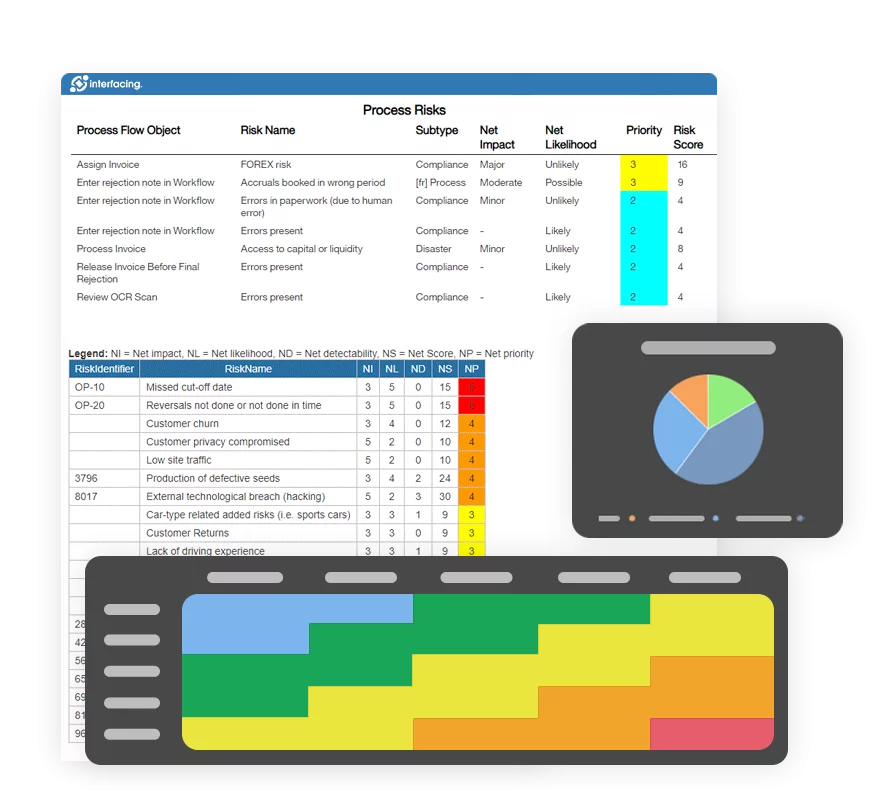
- Risk Management
- Incident Management
- Environmental Health & Safety
- Supplier & Contractor Management
- Product & Supplier Management (SCAR)
- Training Management
- Control Management
- Action Items Management
- Management Review
- FMEA
- Pharmacovigilance
- Data Migration & Integration
 Risk, Governance & Compliance (GRC)
Risk, Governance & Compliance (GRC)- Risk, Governance & Compliance Overview
- Risk & Control Management
- Regulatory Compliance
- Collaboration & Governance
- Data Migration & Integration
- Interfacing Offline App
 Low Code Rapid Application Development (LC)
Low Code Rapid Application Development (LC)- Low Code Automation Platform Overview
- Electronic Web Form Design (eFORMS)
- Database Table Entity Designer
- System Integration Designer
- Design & Manage Tasks
- Design & Manage BPMS Apps
- Custom Rules/Guards/Actions
- Electronic Services
- User Homepage
- BAM (Business Activity Monitoring)
- Custom Dashboard Design
- Data Migration & Integration
 Business Continuity Management (BCM)
Business Continuity Management (BCM)- Business Continuity Management Overview
- Business Impact Analysis
- Disaster Recovery Simulation
- Action Item Management
- Mass Notification Management
- Asset Management
- Interfacing Offline App
 Enterprise Architecture (EA)
Enterprise Architecture (EA) - IndustriesRegulatory ComplianceUse CasesLearning CenterFramework & PracticesIndustries
- Healthcare
- Medical Device Technology
- Life Science, Pharmaceutical
- Aerospace & Defense
- Airlines and Aviation
- Media & Telecommunications
- Government and Military
- Technology
- Energy
- Logistics & Port Operations
- Banking & Capital Markets
- Retail & Consumer
- Consulting
- Education
- Engineering & Construction
- Manufacturing
- Financial Services
- Insurance
- Chemicals
Regulatory Compliance- Regulatory Compliance
- ISO
- ISO 9001 (guide)
- ISO 9001:2026 (preparation)
- ISO 17025
- ISO 27000
- ISO 27001
- ISO27002
- ISO 42001
- EU AI Act
- SOC 2 Type 1 & 2
- Sarbanes Oxley
- GxP
- GRC
- Basel
- Digital Signature
- GDPR
- IFRS
- NIST SP 800-53
- MEL in Aviation
 Use Cases
Use Cases- Quality Management System (QMS)
- Digital Transformation
- Continuous Improvement
- Governance, Risk & Compliance
- Knowledge Management
- System Deployment (ERP, CRM…)
 Learning CenterFramework & Practices
Learning CenterFramework & Practices - AboutCustomer SuccessPartners


Leading AI-Powered
QMS Solutions for Life Sciences & Healthcare
Trusted by Regulated Industries Worldwide
Gartner® Recognized Digital Twin of an Organization (DTO)
ISO, FDA, and GxP Validated Platform
Global Deployments Across Life Sciences, Pharma & Healthcare
Why Modernize Quality & Compliance Now
Align with evolving global standards (ISO 13485, 21 CFR Part 820, GMP/GxP).
Ensure patient safety & data integrity through connected oversight.
Unify fragmented CAPA, audit & training systems.
Accelerate digital transformation while maintaining control.
The Digital Advantage in Healthcare Quality
| Area | Legacy Approach | Interfacing IMS (Outcome) |
|---|---|---|
| Document Control | Manual reviews, disconnected repositories | Automated lifecycle with approval workflows & 21 CFR Part 11 compliance |
| CAPA Management | Reactive follow-ups and manual root-cause logging | AI-assisted root cause, automated notifications, faster closure cycles |
| Audit Readiness | Time-consuming data collection and version hunts | Continuous audit readiness with real-time traceability dashboards |
| Risk Management | Siloed spreadsheets, inconsistent scoring | Integrated risk & control libraries linked to processes and KPIs |
| Regulatory Change Tracking | Reactive updates when regulations change | Proactive impact analysis maps evolving GxP, ISO, FDA changes to SOPs |
| Training & Competency | Manual assignment and tracking | Automated training assignment, alerts, and completion validation |
| Supplier Quality | Email-based communication and fragmented reviews | Centralized supplier management with digital scorecards and workflows |
| Process Visibility | Static flowcharts, disconnected systems | Live Digital Twin of Operations (DTO) with KPI-driven process analytics |
| Data Integrity & Security | Version confusion and limited audit trails | Controlled access, full audit history, and validated data integrity |
| Continuous Improvement | Ad-hoc reviews and siloed feedback | Closed-loop improvement cycles linking NCs, CAPAs, and management reviews |
Core Capabilities
Quality & Document Control
Automate SOP creation, approval, and review cycles.
Risk & Control Management
Identify and mitigate risk proactively.
CAPA & Audit Management
Integrated workflows with AI-suggested root causes.
Regulatory Intelligence
Monitor changes across EU, FDA & ISO.
Training & Competency
Assign and track compliance training automatically.
Archiving & Traceability
Long-term audit-ready document retention.
Research and Development through Manufacturing and Post-market monitoring.
The Life Sciences and Healthcare industries operate under some of the most stringent global regulations, from FDA 21 CFR Part 820 and EU MDR to GxP and ISO 13485.
Ensuring patient safety, product quality, and data integrity requires not just compliance, but continuous improvement powered by data and automation.
Interfacing’s AI-powered Integrated Management System (IMS) gives pharmaceutical, biotechnology, and healthcare organizations a connected platform to manage processes, quality, risk, and compliance, from research and development through manufacturing and post-market monitoring.
The Challenge: Evolving Compliance in a Complex Landscape
The complexity of modern Life Sciences operations continues to grow, distributed sites, outsourced production, supply chain dependencies, and increasingly digital clinical data all demand stronger traceability and oversight.
Traditional document-centric systems and manual processes struggle to keep pace.
Organizations need real-time visibility, closed-loop CAPA management, automated change control, and integrated training tracking to remain compliant and audit-ready while driving efficiency.


The Solution: Digital QMS Powered by AI
Interfacing’s platform unifies all quality and compliance functions into one secure environment, linking SOPs, risks, controls, CAPA, training, audits, and regulatory requirements.
With embedded AI, teams can automatically identify process bottlenecks, assess compliance impact, and predict emerging risks.
Key capabilities include:
AI-Assisted Process Discovery & Mining – uncover inefficiencies and compliance gaps using real event data.
Predictive Quality Analytics – forecast deviations, CAPA delays, or recurring non-conformities before they occur.
Digital Twin of the Organization (DTO) – visualize the entire value chain to understand how process, people, and policy changes impact compliance.
Low-Code Workflow Automation – rapidly digitize and deploy new forms, approvals, and training workflows without coding.
21 CFR Part 11 / Annex 11 Validation Support – ensure data integrity and e-signature compliance.
Learn more about AI for Compliance & Quality.
Designed for Life Sciences & Healthcare Excellence
Pharma: GMP, GxP, and FDA 21 CFR Part 11 compliance.
Medical Devices: ISO 13485 & EU MDR traceability.
Healthcare Providers: Patient safety, operational excellence.

Integrated QMS for GxP and ISO 13485
Built for highly regulated environments, Interfacing’s IMS ensures traceability from design and manufacturing to post-market surveillance, linking every record to its corresponding process and control.
Our digital QMS supports:
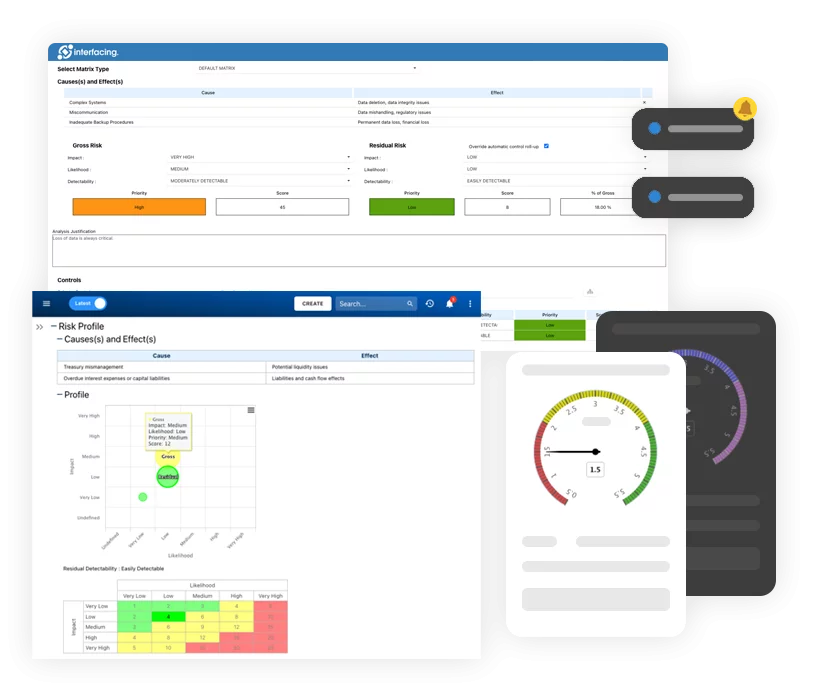
Design & Development Controls
Risk Management & FMEA
Supplier Qualification & Audits
Training & Competency Management
CAPA and Non-Conformance
Complaint Handling & Vigilance Reporting
Document & Change Control
See how the system aligns with ISO 13485 Compliance for Medical Device Companies and FDA 21 CFR Part 820 Requirements.
Data-Driven Decision Making
Beyond compliance, AI enables transformation.
By connecting process data with operational metrics, organizations gain the intelligence to:
Improve quality review cycle times
Reduce human error and rework
Detect cross-functional process dependencies
Increase right-first-time outcomes
Strengthen regulatory readiness through audit dashboards and impact analysis
Explore the broader context of GxP Risk Management & Compliance.

Steps to ISO 45001 Certification
Achieving certification is a multi-phase journey. Here’s how organizations succeed:
1: Gap Analysis
Use digital assessments to evaluate how current safety practices compare to ISO 45001 standards. AI tools can accelerate documentation audits and identify high-risk process gaps.
2: Define Scope and Objectives
Clearly outline operational boundaries and measurable OH&S goals, ensuring alignment with strategic business targets.
3: Leadership Commitment and Planning
Assign top-level responsibility, establish a safety policy, and conduct stakeholder consultations. Interfacing supports role-based access and approval workflows to manage this process.
4: Hazard Identification and Risk Assessment
Develop a proactive risk matrix using historical data, predictive AI tools, and frontline input. Integrate this directly into your SOPs.
5: Operational Controls and Training
Implement safety protocols, emergency procedures, and competency-based training modules. With Interfacing, you can automate training assignments and track completions.
6: Performance Evaluation and Internal Audit
Measure KPIs, conduct internal audits, and perform root cause analysis on incidents. Real-time dashboards help management review OH&S performance consistently.
7: Certification Audit
Engage an accredited third-party auditor. Interfacing’s audit-ready documentation, traceability logs, and eSignatures ease this process.

Common Pitfalls to Avoid
Many organizations struggle with implementation due to:
Inadequate Top Management Engagement
Without leadership buy-in, ISO 45001 becomes a checklist exercise. Interfacing helps enforce executive accountability via workflow governance and digital signoffs.
Overlooking Non-Employee Risks
Contractors, suppliers, and temporary workers are often excluded from OH&S planning. This introduces major blind spots.
Poor Documentation and Change Control
Stale documents, unapproved updates, and missing records are major causes of audit failure. Interfacing’s document lifecycle tools ensure version control and full audit trails.
Neglecting Mental Health and Ergonomics
Modern safety includes psychosocial risks. AI can monitor trends like absenteeism, burnout indicators, or repetitive strain injuries to improve overall workplace health.
How Interfacing Helps
Interfacing’s Integrated Management System (IMS) acts as the digital backbone for life sciences quality operations — delivering end-to-end control, visibility, and automation.
Unify your quality ecosystem: link documents, processes, risks, and training in one central repository.
Prove compliance: full traceability from procedure to evidence with electronic signatures and automated audit logs.
Accelerate innovation: use AI insights to shorten cycle times, identify recurring issues, and automate quality improvements.
Simplify validation: Interfacing’s validated environment supports 21 CFR Part 11 / Annex 11 standards.
Request a demo of the AI-Integrated Management System to see how it can transform your quality operations.

Ensure Process & Quality Governance
Interfacing’s Enterprise Process Center® (EPC) allows you to define, document, and enforce ISO 9001 quality controls organization-wide. Policies, procedures, and SOPs are directly tied to processes and roles—ensuring your QMS is embedded in everyday operations with full transparency. .

Eliminate Manual Errors with AI-Driven QMS
Standardizing documentation and workflows within EPC removes the need for spreadsheets and disconnected systems. Our AI flags outdated procedures, suggests process improvements, and helps maintain version control—reducing the risk of non-compliance and audit issues.

Gain Full Audit Readiness & Traceability
EPC provides real-time traceability across all quality processes—from document approvals to CAPA workflows. With automated audit trails and visual process maps, you’re always ready for certification audits and internal reviews, no last-minute scrambling required

Improve Operational Efficiency Without Sacrificing Compliance
Interfacing’s QMS automates routine tasks such as change control, training sign-offs, and document updates. This not only frees up valuable staff time, but also ensures consistent adherence to ISO 9001 guidelines across all departments and geographies.

Reduce the Cost of Quality Compliance
With centralized process governance and AI-enhanced workflows, EPC reduces the overhead of maintaining your QMS. From smarter audit prep to fewer non-conformities, organizations save time, cut errors, and accelerate their path to ISO 9001 certification.

Build a Culture of Continuous Improvement
ISO 9001 is about more than passing audits—it’s about evolving your organization. Interfacing’s platform helps identify quality gaps, track corrective actions, and promote ongoing learning and accountability—turning compliance into a competitive advantage.
Why Choose Interfacing?
With over two decades of AI, Quality, Process, and Compliance software expertise, Interfacing continues to be a leader in the industry. To-date, it has served over 500+ world-class enterprises and management consulting firms from all industries and sectors. We continue to provide digital, cloud & AI solutions that enable organizations to enhance, control and streamline their processes while easing the burden of regulatory compliance and quality management programs.
To explore further or discuss how Interfacing can assist your organization, please complete the form below.

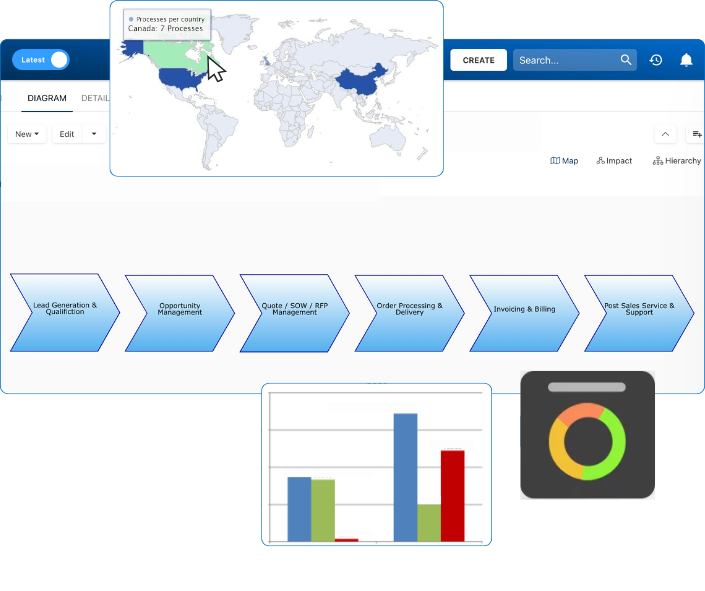
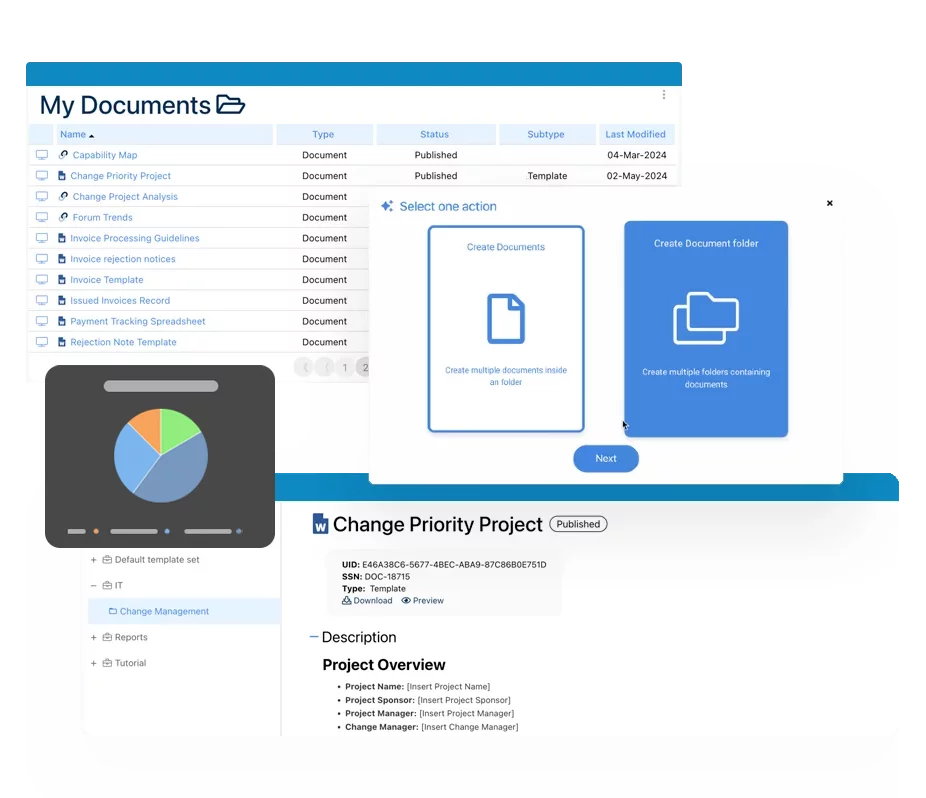
Documentation: Driving Transformation, Governance and Control
• Gain real-time, comprehensive insights into your operations.
• Improve governance, efficiency, and compliance.
• Ensure seamless alignment with regulatory standards.

eQMS: Automating Quality & Compliance Workflows & Reporting
• Simplify quality management with automated workflows and monitoring.
• Streamline CAPA, supplier audits, training and related workflows.
• Turn documentation into actionable insights for Quality 4.0

Low-Code Rapid Application Development: Accelerating Digital Transformation
• Build custom, scalable applications swiftly
• Reducing development time and cost
• Adapt faster and stay agile in the face of
evolving customer and business needs.
AI to Transform your Business!
The AI-powered tools are designed to streamline operations, enhance compliance, and drive sustainable growth. Check out how AI can:
• Respond to employee inquiries
• Transform videos into processes
• Assess regulatory impact & process improvements
• Generate forms, processes, risks, regulations, KPIs & more
• Parse regulatory standards into requirements

Request Free Demo
Document, analyze, improve, digitize and monitor your business processes, risks, regulatory requirements and performance indicators within Interfacing’s Digital Twin integrated management system the Enterprise Process Center®!
Trusted by Customers Worldwide!
More than 400+ world-class enterprises and management consulting firms