Most organizations rely on CAPA systems to manage quality events, yet recurring issues continue to surface across operations. Investigations are completed, actions are assigned, and records are closed, but the same problems reappear. The issue is not a lack of data. It is the absence of operational context needed to explain why those problems occur in the first place.
CAPA Systems Are Designed to Record, Not Explain
Corrective and Preventive Action (CAPA) systems are a foundational component of quality management. They are designed to capture deviations, assign actions, and document resolution activities in a structured and auditable way.
On paper, this appears sufficient. Organizations can demonstrate that issues are tracked, investigated, and closed in compliance with regulatory expectations.
But this creates a false sense of control.
Most CAPA systems answer what happened:
- A deviation occurred.
- A non-conformance was identified.
- An investigation was conducted.
- Corrective actions were assigned.
What they rarely answer is:
Why did this happen within the operation?
Where in the process did the breakdown occur?
What conditions allowed the issue to persist or repeat?
Without these answers, CAPA becomes a documentation exercise rather than a mechanism for improvement.
The Root Cause Problem Is Not Analytical, It Is Structural
Many organizations assume that weak root cause analysis is the result of poor methodology. They introduce tools such as 5 Whys, fishbone diagrams, or comparative analysis techniques.
These methods are valuable, but they depend on one critical input that is often missing, a clear understanding of how the process actually operates.
When teams do not have visibility into:
- end-to-end process flows
- role responsibilities across departments
- system interactions and dependencies
- control points and decision logic
Their analysis becomes speculative.
Investigations rely on interviews, assumptions, and fragmented data sources. Root causes are often described at a surface level, such as “human error” or “process not followed,” without identifying the systemic conditions behind the issue.
This is not a failure of analytical skill. It is a failure of operational context.
Disconnected Systems Create Disconnected Investigations
In most organizations, the data required to understand a quality issue exists across multiple systems.
Process documentation is stored in one platform.
Training records exist in another.
Audit findings are tracked separately.
Risk registers are maintained independently.
Operational workflows are managed elsewhere.
When a CAPA is initiated, teams must manually assemble information from these sources to reconstruct what happened.
This introduces several problems.
First, investigations become time-consuming and inconsistent. Each team builds its own version of the operational narrative.
Second, critical relationships are often missed. A deviation may be linked to a training gap, a process variation, or a control failure, but these connections are not easily visible.
Third, recurring issues remain undetected at a systemic level. CAPA systems track individual events, but they rarely reveal patterns across processes.
As a result, organizations treat symptoms repeatedly without addressing the underlying operational design.
CAPA Without Process Context Leads to Repetition
One of the clearest indicators of CAPA failure is recurrence.
The same type of deviation appears across different teams.
Corrective actions are implemented but fail to prevent future issues.
Audit findings reappear despite prior remediation efforts.
This happens because CAPA actions are often applied locally rather than systemically.
A procedure may be updated.
Additional training may be assigned.
A control may be reinforced.
But the broader process structure, including handoffs, dependencies, and decision logic, remains unchanged.
Without understanding how the process operates as a whole, organizations cannot identify where risk is embedded within execution.
CAPA becomes reactive by design.
From Event Tracking to Operational Understanding
To move beyond reactive CAPA management, organizations need to shift their perspective.
Instead of treating CAPA as a standalone quality function, it must be connected directly to the operating model.
This means linking quality events to:
- the processes where they occur
- the roles responsible for execution
- the controls designed to prevent failure
- the data that reflects real operational behavior
When CAPA is grounded in this context, investigations change fundamentally.
Teams can trace how a deviation originated within a process flow.
They can identify whether the issue is caused by design, execution, or control failure.
They can assess the impact of the issue across related processes and systems.
Root cause analysis becomes evidence-based rather than assumption-driven.
The Role of AI-Assisted Insight in CAPA
AI-assisted capabilities can further enhance CAPA effectiveness, but only when applied within a governed operational framework.
When processes, risks, controls, and quality events are connected, AI can help identify patterns that are difficult to detect manually.
For example:
- Recurring deviations across similar process steps
- Correlations between training gaps and quality incidents
- Control failures linked to specific operational conditions
However, AI does not replace the need for structure. It depends on a well-defined operating model to generate meaningful insights.
Without that foundation, AI simply accelerates analysis on incomplete or disconnected data.
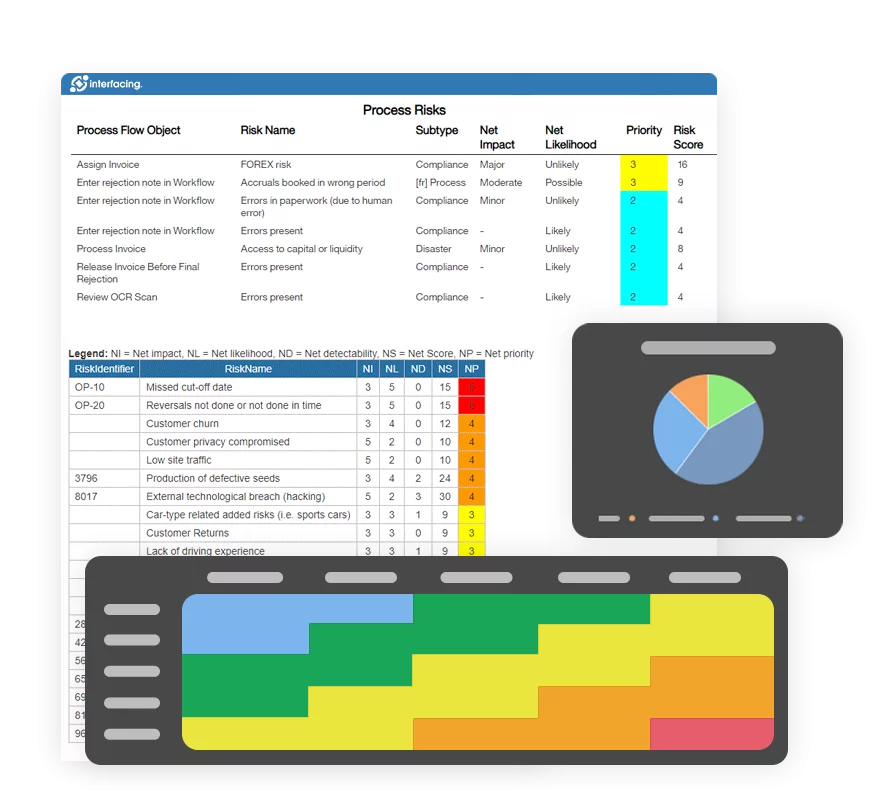
How Interfacing Enables True Root Cause Visibility
Interfacing Technologies Corporation enables organizations to move beyond traditional CAPA systems by connecting quality management directly to the operating model.
Through its integrated management system, CAPA events are linked to processes, roles, controls, and related operational data within a single environment.
This allows organizations to understand not just that an issue occurred, but how and why it emerged within the broader operational context.
Teams gain the ability to trace deviations across process flows, identify systemic weaknesses, and implement corrective actions that address root causes rather than symptoms.
This approach transforms CAPA from a reactive tracking mechanism into a driver of continuous improvement.
Final Thoughts
CAPA systems do not fail because they lack data. They fail because they lack context.
Capturing issues, assigning actions, and closing records are necessary for compliance, but they are not sufficient for operational improvement.
Organizations that continue to experience recurring issues are not facing a documentation problem. They are facing a visibility problem.
By connecting CAPA to the operating model, organizations can move from reactive problem management to true root cause understanding.
Only then can corrective actions deliver lasting impact.
Why Choose Interfacing?
With over two decades of AI, Quality, Process, and Compliance software expertise, Interfacing continues to be a leader in the industry. To-date, it has served over 500+ world-class enterprises and management consulting firms from all industries and sectors. We continue to provide digital, cloud & AI solutions that enable organizations to enhance, control and streamline their processes while easing the burden of regulatory compliance and quality management programs.
To explore further or discuss how Interfacing can assist your organization, please complete the form below.

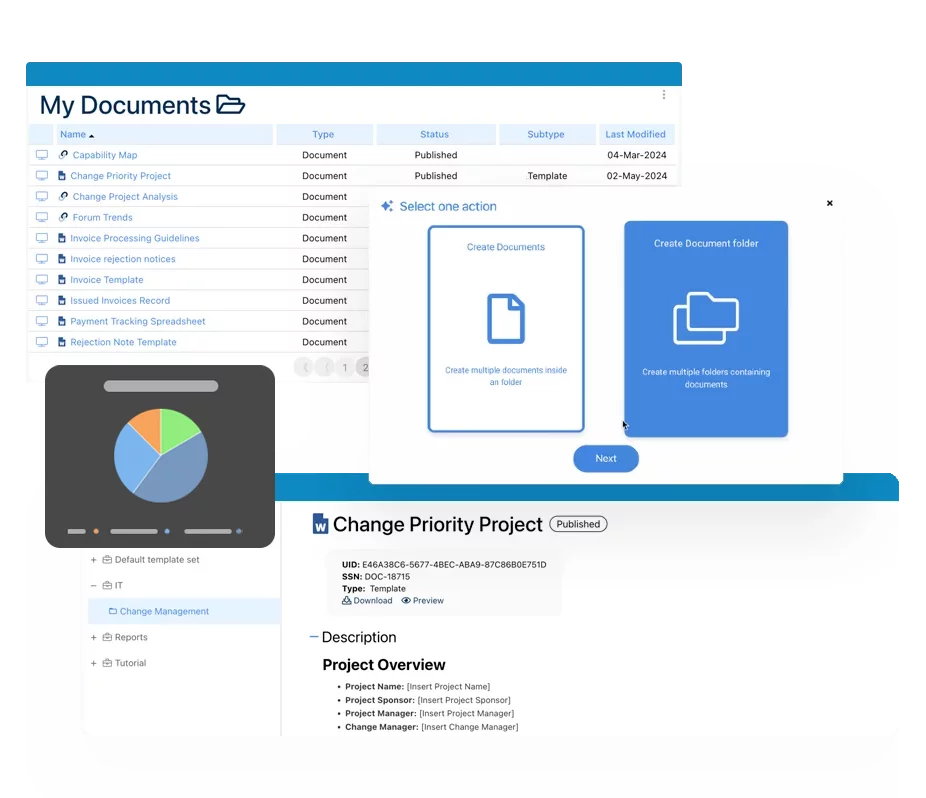
Documentation: Driving Transformation, Governance and Control
• Gain real-time, comprehensive insights into your operations.
• Improve governance, efficiency, and compliance.
• Ensure seamless alignment with regulatory standards.

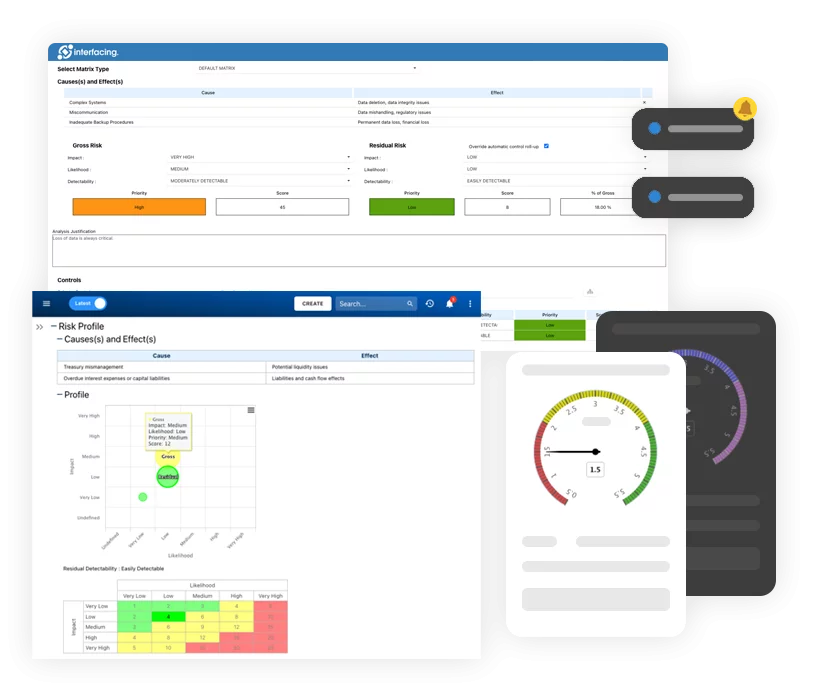
eQMS: Automating Quality & Compliance Workflows & Reporting
• Simplify quality management with automated workflows and monitoring.
• Streamline CAPA, supplier audits, training and related workflows.
• Turn documentation into actionable insights for Quality 4.0

Low-Code Rapid Application Development: Accelerating Digital Transformation
• Build custom, scalable applications swiftly
• Reducing development time and cost
• Adapt faster and stay agile in the face of
evolving customer and business needs.
AI to Transform your Business!
The AI-powered tools are designed to streamline operations, enhance compliance, and drive sustainable growth. Check out how AI can:
• Respond to employee inquiries
• Transform videos into processes
• Assess regulatory impact & process improvements
• Generate forms, processes, risks, regulations, KPIs & more
• Parse regulatory standards into requirements

Request Free Demo
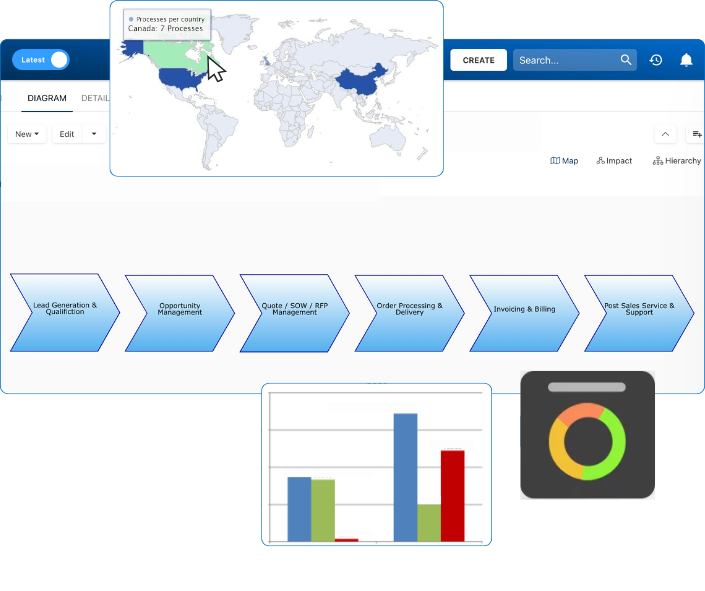
Document, analyze, improve, digitize and monitor your business processes, risks, regulatory requirements and performance indicators within Interfacing’s Digital Twin integrated management system the Enterprise Process Center®!
Trusted by Customers Worldwide!
More than 400+ world-class enterprises and management consulting firms